| Vevo News Share Your Story - February 2016 |
 |
 |
Using the VevoLAZR to characterize the tumor microenvironment and develop innovative contrast agents. Submitted by Dr. Giuseppe Ferrauto |
Molecular Imaging is emerging as an innovative and multidisciplinary field of research aimed at visualizing and quantifying the signature of diseases, making it possible for an earlier and more precise diagnosis. The simultaneous application of different imaging modalities, each of them endowed with a different portfolio of applications and advantages, can enhance the information about the diseases being investigated. In this context, photoacoustic imaging (PAI) is attractive because it merges the advantages of ultrasound with the intrinsic optical absorption properties of molecules naturally occurring in tissues or exogenously administered. At the Molecular Imaging Centre at the University of Torino- Italy (under the Supervision of Prof. Silvio Aime), there is a great deal of experience in the development of contrast agents for different imaging modalities, and in the development of innovative procedures for the extensive characterization of diseases, especially cancer. In this framework, the application of the VevoLAZR system in our group is devoted to two main purposes: i) the visualization and quantification of vascular volume and oxygen saturation in cancerous regions and ii) the characterization of innovative supramolecular PAI contrast agents for theranostic applications. |
| Dr. Giuseppe Ferrauto |
__________________________________________________________________________________________________ Giuseppe Ferrauto is a post-doctoral researcher at the Molecular Imaging Center- University of Torino (It) in Prof. Silvio Aime’s group. His research is mainly focuses on the development of innovative nano/micro-sized contrast agents andprocedures (for MRI and PAI) for the molecular characterization of the tumor microenvironment. He received the EMIM (European Society for Molecular Imaging ) Best PhD Thesis Award in 2014 and the Vevo Travel Awards for Cancer Track in 2015 . |
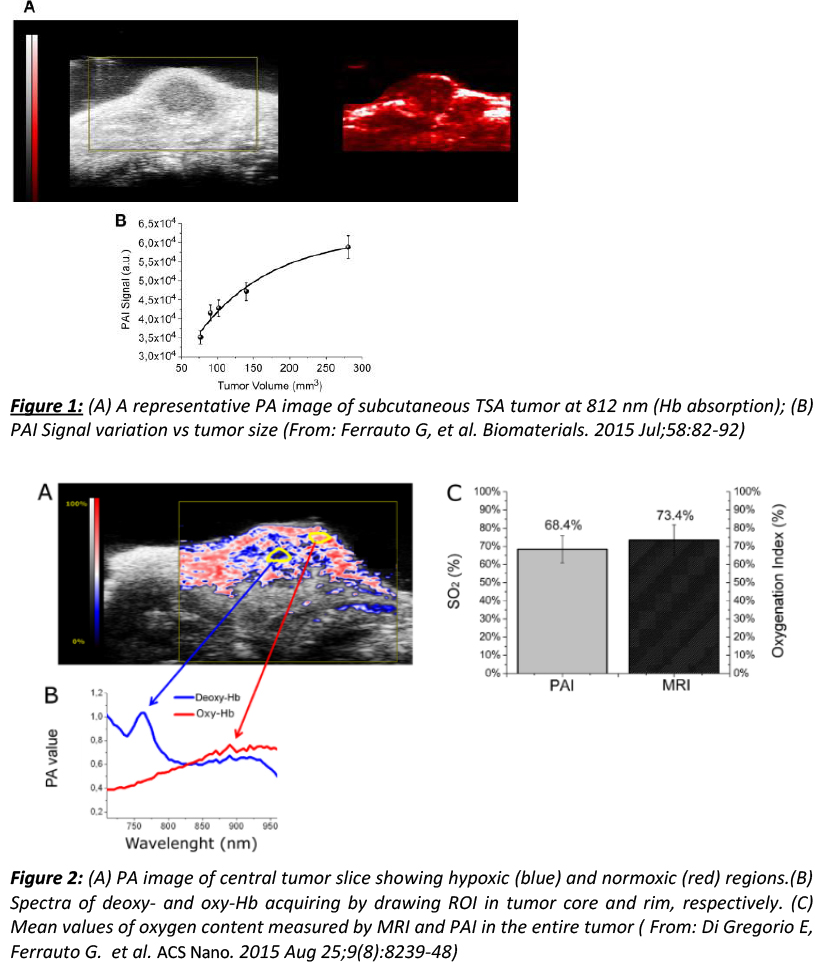 |
 |
As far as the first application, our group is investigating changes occurring in the vascular network (vascular volume and hypoxia) during tumor growth in breast and prostate cancer models. The aim is to correlate these data with tumor aggressiveness, and to develop innovative anti-cancer therapies and strategies to monitor their effects in vivo. PAI has played an important role in validating innovative MRI tools based on the incorporation of Gd-complexes inside red blood cells, that have been recently developed in our center, with the aim of assessing tumor vascular volume and hypoxia (Ferrauto G, et al. Biomaterials. 2015 Jul;58:82-92 and Di Gregorio E, Ferrauto G. et al. ACS Nano. 2015 Aug 25;9(8):8239-48). As far as the second application, PAI is used for the development of innovative theranostic PAI contrast agents. In particular, we are exploring the feasibility of using biocompatible nanosized systems (liposomes, polymeric vesicles, etc...) loaded with cyanine dyes as PAI agents for application in molecular imaging studies. Figure 1: (A) A representative PA image of a subcutaneous TSA tumor at 812 nm (Hb absorption); (B) PAI signal variation vs tumor size (From: Ferrauto G, et al. Biomaterials. 2015 Jul;58:82-92) Figure 2: (A) PA image of central tumor slice showing hypoxic (blue) and normoxic (red) regions.(B) Spectra of deoxy- and oxy-Hb acquired by drawing ROI in tumor core and rim, respectively. (C) Mean values of oxygen content measured by MRI and PAI in the entire tumor (From: Di Gregorio E, Ferrauto G. et al. ACS Nano. 2015 Aug 25;9(8):8239-48) |